This article is cross-posted from the Wild Animal Initiative website. The original is here. It is also available on the EA Forum here.
Summary
In order to determine which conditions provide the best overall quality of life for nonhuman animals, it is important to be able to measure their cumulative welfare experience. The ideal measure of cumulative welfare would be comprehensive, objectively measurable, and easy to transfer across species; however, existing approaches fall far short of this ideal. Recent academic work has suggested that measures of biological ageing could provide a highly promising alternative measure of cumulative welfare, which comes much closer to meeting these ideal goals.
Here, I review the existing empirical support for the use of biomarkers of ageing as a measure of cumulative welfare, discuss the prerequisites of applying the method, and explore a number of important caveats that may limit its applicability. Many of these caveats are particularly applicable to the study of wild-animal welfare, though some may also be important in domesticated contexts.
Overall, despite some important potential weaknesses, biomarkers of ageing are likely to represent an important step forward in the assessment of cumulative animal welfare, which could potentially help resolve some important long-running uncertainties and disputes in the animal-welfare movement. Wild Animal Initiative recommends that both researchers and funders take note of these new techniques, and consider how best they can develop them further or apply them in their own domains of expertise.
We need a good way of measuring cumulative animal welfare
While single experiences can be acutely positive or negative, what matters more from a welfare perspective is the lasting cumulative impact of these experiences.
— Bateson & Pourier 2019
Over the course of their life, an animal will undergo various positive and negative experiences. Some of these experiences will primarily affect welfare in an acute, short-lived manner, while others will have significant long-lasting welfare effects. The overall welfare state of an animal will be determined by the cumulative effect of all the experiences they have faced in the course of their life to date.
In order to improve animal welfare, we need to know what affects it. In order to improve animal welfare effectively, we need to know which factors have the greatest total effect on animals’ cumulative wellbeing. Actually measuring cumulative wellbeing, however, is highly challenging, and our existing methods for doing so frequently rely on crude proxies or error-prone anthropomorphic judgements. To make matters worse, the welfare effect of a given experience is frequently complex, species-specific, and non-obvious to humans:
- Species of fish which are naturally solitary exhibit stress indicators upon overcrowding, while schooling fish become stressed if raised at unnaturally low densities .
- Exposure to environmental ultrasound frequencies that are undetectable to humans causes depression-like symptoms in laboratory rats and mice .
- The flickering of some fluorescent lights at frequencies above the human flicker-fusion rate raises stress-hormone levels in starlings .
- The stress-hormone levels induced in captive-bred lizards by different experimental procedures can differ dramatically from how stressful human observers think the procedures are, with one controversial procedure found to generate much lower levels of stress hormone than the “less-stressful” procedure that replaced it .
Which specific stimuli are important for an animal’s welfare is therefore very difficult to predict a priori, and the use of anthropomorphism to make these predictions is fraught with danger.
Even if it is possible to determine that a given experience is acutely positive or negative for an animal, it is not obvious how to convert these acute measurements into measurements of lasting impacts on welfare. Experiences with similar acute effects may have dramatically differences on long-run wellbeing: one stressor may involve temporary pain or result in a temporary spike in stress-hormone levels but have no long-term effects, while another might significantly contribute to long-term stress levels. In some cases, the short- and long-term welfare impacts of an exposure may even be of opposite sign: some acute stresses can be beneficial in the long-term, while the short-term pleasure of sugary food might be outweighed by the long-term pain caused by tooth decay and weight gain . Worse, the relationship between short-term and long-term welfare impacts is likely to vary significantly based on species, subspecies, chronological age, past experiences, and individual genetic variation, making the long-term welfare effect of an acute experience even more difficult to predict.
A good measure of cumulative affective experience is therefore vital to the study of animal welfare. However, developing a cumulative-welfare metric which is sensitive, easy-to-measure, captures all or almost all of the relevant effects on welfare, and can be transferred with relative ease across species has proven to be highly challenging . Existing measures include chronic physiological markers such as resting stress-hormone levels or bodyweight, acute behavioural measures such as the presence of stereotypies, and high-level behavioural measures such as depressive symptoms or cognitive biases ; other measures combine multiple different sources of evidence, often alongside acute welfare indicators and/or subjective welfare assessments by a trained practitioner. However, all of these methods have serious drawbacks: physiological measures often lack sensitivity and specificity (i.e. they do not always reliably correlate with affective state), stereotypies are highly species-specific and often difficult to interpret, and the behavioural tests required to measure cognitive biases and mood must be developed and validated independently for each species and often require extensive animal training. Combined measures, meanwhile, rely on difficult-to-test assumptions about how different metrics should be combined and weighted to assess overall cumulative wellbeing.
In the ideal case, the many and varied inputs into cumulative animal welfare would be captured by a single, objectively measurable metric, which could then be used as a single readout of cumulative wellbeing for many different species of animals. In the rest of this article, I will review and discuss the evidence supporting a new, surprisingly good candidate for this “objectively measurable common currency” of wellbeing: biomarkers of the ageing process.
Biological ageing and cumulative animal welfare
As we get older, our bodies decay. In various ways, our cells and tissues progressively accumulate increasing levels of damage and dysregulation, leading at the whole-organism level to a decline in functionality, an increase in mortality, and a decrease in reproductive output . These deteriorative processes, which occur in broadly similar ways in many different animal species , are collectively known as ageing.
Among researchers studying the biology of ageing, it is well-known that simple chronological age is a less-than-perfect measure of the aspects of ageing we tend to care about. In humans, individuals of the same chronological age often differ substantially, both in how old they appear to others (degree of graying, wrinkles, stooping, etc.) and in their age-related health outcomes . It is therefore useful to separate the concepts of “chronological age” (time since conception or birth) and “biological age” (degree of age-related change/deterioration in appearance, health, or functionality) . So-called “biological ageing clocks”, which incorporate a variety of different types of biological data, are an active area of research in the study of human ageing, and the best such clocks can predict health and other outcomes much more accurately than chronological age alone .
The biological age of an individual depends on their chronological age, genetic background, and environmental history: depending on their genes and experiences, two individuals of the same chronological age can differ substantially in their biological age. A striking example of this is smoking, which produces a variety of ageing-like symptoms and has recently been shown to substantially increase biological age in young humans . However, biological age is affected by a wide variety of chemical, psychological and social stimuli, many of which are also known to have important effects on an individual’s wellbeing. To take just one example, a wide variety of negative experiences (including anxiety, depression, childhood trauma, chronic pain, and various forms of stress) are associated with reduced telomere length in humans, while positive lifestyle factors are associated with longer telomeres . Despite important differences in telomere biology between species, a variety of stress manipulations (including social isolation, sleep disruption, injection of stress hormones, and crowding) have also been found to accelerate telomere attrition in various nonhuman animals, including wild and laboratory mice, chickens, starlings, and various other bird species .
In addition to this and other empirical data, there are good theoretical reasons to expect the rate of biological ageing to correlate with the cumulative affective experience of an individual . Evolutionarily speaking, the affective state of an experience serves to motivate an animal to seek or avoid similar experiences; hence, affectively negative experiences tend to be those that reduce animal’s fitness, while affectively positive experiences tend to be those that increase it. An important way in which an experience can decrease fitness is by causing or contributing to some sort of damage or dysregulation in the body; hence, experiences which contribute to damage or dysregulation will typically be perceived as aversive to the organism, while those that prevent or reverse damage (or have no effect on damage but are positive for some other reason) will be perceived as attractive. In general, therefore, there is good reason to expect physiologically-damaging experiences to be affectively negative, and vice versa , with a similar connection between physiologically-protective experiences and positive affect. Since ageing is characterised by the progressive accumulation of various forms of physiological damage, this suggests that there is good reason to expect the affective valence (i.e. the positivity or negativity) of an experience to also generally correlate with its effect on biological age.
There are, therefore, both empirical data and theoretical arguments suggesting a relationship between cumulative affective experience and ageing. If this relationship exists, the biological age of an individual relative to their chronological age could be used to assess that individual’s cumulative welfare experience up to that point. If further testing bears this out, there are several reasons to expect biological age to be a particularly valuable tool for assessing wellbeing:
- It is highly general, including all (or almost all) causes of stress and wellbeing experienced by the animal, including those not obvious or perceptible to humans
- It is cumulative, giving a readout of the total affective history of an individual
- It is objectively measurable, with well-established biomarkers already known for a number of species
- Finally, it is plausibly relatively phylogenetically neutral: as ageing is a general phenomenon shared by very many species, whose measurement does not generally rely on anthropomorphic judgements, it can potentially be used to investigate welfare in many different animal groups.
Given these potential advantages, how might we go about actually measuring biological age?
Measuring biological age in non-human animals
In any given species, the ageing process will manifest itself in a plethora of different phenotypes, many of which can be used to try to quantify biological ageing. In humans, a wide variety of biological readouts have been used as biomarkers of ageing, including telomere length and attrition rate, DNA methylation patterns, gene expression profiles, changes in neuroanatomy, proteomic and metabolomic changes, and various composites of clinically relevant symptoms . While many of these biomarkers predict health outcomes better than chronological age, they often reflect different aspects of the ageing process and do not always correlate well with one another . Combining different measures (e.g. with machine-learning-based prediction tools) can overcome these problems and improve the ability of a biological ageing measure to predict health outcomes .
As different biomarkers of ageing track different aspects of the ageing process, they may differ in how well they measure cumulative subjective wellbeing. A good biomarker of cumulative experience should provide a single, continuous, easy-to-measure readout that responds in opposite directions to positive and negative affective experiences in a cumulative and dose-dependent manner . Different biomarkers will also differ in the money, expertise and time required to obtain good measurements, and in the ease with which they can be validated in a new context. As nonhuman animals, unlike humans, cannot explicitly self-report their subjective experience, validation of a new potential biomarker’s relationship with wellbeing is much more challenging in these species; as a result, the ease and reliability with which a biomarker can be transferred between species is an additional important consideration when choosing how to measure biological age in an animal-welfare context.
Overall, I would expect more complex and multi-modal measures to provide a more accurate, precise and thorough measure of biological age and so give a better idea of an animal’s cumulative experience. On the other hand, I would expect these complex measures to be more expensive and time-consuming to obtain for each individual, and less transferable between species.
For some widely-used farmed species and experimental model organisms, it may well be worth developing sophisticated species-specific methods of measuring biological age and hence cumulative welfare; however, in contexts where resources are highly limited and/or the number of species of concern is large, cost and transferability concerns are likely to mitigate in favour of simpler, faster, cruder measures . Bateson & Pourier suggest telomere length and hippocampal volume as two metrics that are simple, well-defined, and likely to retain validity across a wide variety of vertebrate species; further work may reveal other promising candidates. However, when using such very simple readouts of biological age, it is important to remember that they may give a significantly more partial and inaccurate reading than more sophisticated measures, and to seek to develop such improved measures where feasible.
A concrete example: the welfare effect of crowding on farmed fishes
Many farmed fish are kept at very high densities, in a manner which often appears to be detrimental to their welfare . The potential welfare effects of crowding are many and varied, including social stresses, reduced water quality, and increased disease transmission. However, the actual welfare effect of a given level of crowding will vary between fish populations, depending on the level of crowding and water quality they are adapted to cope with, the robustness of their immune systems, and whether they have been vaccinated, among other factors. It would be useful to measure the total cumulative welfare effect of different crowding regimes on different species, and to know the degree to which other interventions such as vaccination mitigate any crowding-induced welfare reduction. Biological age provides an ideal means of addressing these questions.
To apply this method, we would need some sort of biological ageing clock for each of the fish species of interest, as well as some way of keeping track of the chronological age of each individual. Once these two methods are in place for each species, the experiment is simple: simply raise populations of fish at different levels of crowding, sample the biological ages of individuals in each population, then compare the biological ages of chronological-age-matched individuals from different conditions. Those populations exhibiting the highest biological age relative to their chronological age would be taken as experiencing the lowest levels of cumulative welfare.

Plots showing speculative results of a hypothetical experiment investigating the effect of crowding on fish welfare, indicating that biological age accumulates slowest (indicating highest welfare) at low densities for solitary fish and middling densities for schooling fish, with a positive welfare effect of vaccination. The shape of the curves is arbitrary.
To make up some totally speculative results, these biological ageing data might indicate that naturally solitary fish exhibit significant welfare declines from any level of crowding, while schooling fish have a preferred crowding level and experience reduced welfare above or below this level. These welfare declines may or may not be abrogated by interventions such as vaccination, better water filtration, or changes in feeding schedule. By collapsing all (or most) of an animal’s welfare experience into a single, objective, cumulative measure, many different experiments of this kind could be performed quickly and efficiently, providing a more comprehensive picture of the welfare effects of crowding on fish wellbeing.
While the idea that crowding is bad for fish welfare may not be particularly controversial, this approach would allow researchers to empirically quantify how bad that effect is compared to other aspects of a farmed fish’s life, and assess the efficacy of different interventions (such as vaccination or water oxygenation) for mitigating that welfare impact. The same technique could be applied to help resolve active empirical controversies in the animal welfare movement, such as the relative welfare levels of caged vs cage-free chickens, the relative importance of water oxygenation in the welfare of farmed fishes, or the net welfare impact of predator reintroduction on prey species.
Limitations and caveats
While I was initially sceptical about the applicability of biological ageing markers as measures of animal welfare, I have generally been convinced that this represents a novel and important advance in the field. Nevertheless, there are a number of important limitations or difficulties I anticipate in actually applying the method, which I think it is important to be aware of and mitigate where possible. I have divided these limitations into two categories: contexts where the prerequisites of applying the method may be difficult or expensive to obtain, and contexts where the link between ageing and welfare may be weakened or broken entirely.
Difficulties in application
The need for biological ageing clocks
In order to use biological ageing as a cumulative welfare measure in a given species, we need some kind of biological ageing measure for that species. As discussed above, these range in complexity from simple metrics like telomere length to highly complex machine-learning-based predictors, and there is likely to be a tradeoff between the accuracy and comprehensiveness of a measure on the one hand and its affordability and transferability on the other. As funding is very limited in many animal-welfare contexts, it is likely that simpler, cheaper metrics that can be transferred between species with relatively little validation will be preferred; however, it is important to remember that these may only provide a partial measure of biological age.
The best biomarkers of ageing to use for these experiments will depend on the species being tested and its relationship to other well-validated model systems. In many vertebrate species, and probably most mammals, the markers established in humans and laboratory mice are likely to be the best option. Conversely, in species very distantly related to humans the validity of these markers may be limited: insects, for example, are largely post-mitotic in their adult form (limiting the usefulness of telomere attrition as a biomarker) and have very different neuroanatomy from vertebrates (preventing the use of hippocampal volume). If it is desirable to apply these ageing-based methods to assess welfare in these species, alternative biomarkers (such as accumulation of fluorescent advanced glycation end products in Drosophila ) will need to be developed and validated as welfare measures, substantially increasing the upfront cost.
Experimental controls
While biological ageing is a promising measure of cumulative welfare, it is important to remember that welfare is not the only thing affecting biological age. Most obviously, chronological age has a very strong effect on biological age, and studies should always compare age-matched individuals when possible. Genetic variability is also an important factor: many species exhibit substantially different lifespans in different populations, and polymorphisms within a population can also have a substantial effect. As a result, ageing-based welfare measures will be most reliable in contexts where all individuals are genetically homogeneous, or at least where there is no systematic difference in genetic composition between different experimental groups. Finally, there should of course be as little systematic difference in environment as possible between the groups being compared, other than whatever exposure is being investigated for its welfare effects.
Of these control requirements, the need for genetic comparability between experimental groups is the most frustrating, as it appears to exclude a lot of factors that are widely thought to be important for animal welfare, particularly in domesticated contexts where animals’ genotypes have been substantially modified by humans through selective breeding. On the face of it, biological-ageing methods seem to be unable to address this, as the groups being compared are not genetically comparable. However, given the importance of genetic effects on welfare in many contexts, any way to overcome this limitation would be very useful, and further investigation on this front seems quite valuable in expectation.
Limitations to validity
Death and other acute events
In many cases, a large portion of the suffering experienced by an animal, whether domesticated or wild, is suspected to take place acutely at the moment of death. If this death is sufficiently gruesome, the suffering so engendered could potentially outweigh the entire net welfare of a life that is otherwise worth living. Despite its importance, however, it seems unlikely that the suffering entailed by dying would be adequately reflected in ageing-based measures of cumulative welfare. For one thing, there would be no opportunity for the negative experiences associated with the animal’s death to be reflected as a subsequent increase in the rate of biological ageing; for another, the extensive physiological damage resulting from death would prevent an accurate postmortem assessment of biological age in many cases. Hence, an important limitation of ageing-based methods of measuring welfare may be their inability to incorporate the affective experience of dying.
A similar concern might apply, albeit to a much lesser extent, to other highly acute exposures, i.e. those with large but short-lived effects on welfare. Many of these will be reflected in ongoing cumulative welfare to some extent (e.g. as physical or psychological trauma in the case of negative events), but it’s not clear to me that the cumulative readout of welfare given by ageing biomarkers will always incorporate them adequately. More research may be needed here.
Differences between juveniles and adults
In both wild and domesticated contexts, many of the animals of greatest concern from a welfare perspective are juveniles who have not yet reached reproductive maturation. These immature individuals are often much more numerous than adults, and have less chance to accumulate positive experiences to outweigh the pain of dying.
For animals that die shortly after birth or hatching, the cumulative welfare of their lives is likely to be dominated by the affective experience of dying, and this experience will not be adequately reflected in biological ageing markers. Ageing-based approaches therefore seem unable to effectively address the welfare of these individuals; on the other hand, it seems likely that any measure of cumulative welfare will run into the same problem.
The situation for individuals that have relatively long lives as juveniles is more complex. In many species, juvenile and adult individuals differ substantially in their biology, and the question of whether juveniles are “ageing” is somewhat fraught. There are certainly dramatic changes taking place over the course of development, some of which could be interpreted as an accumulation of damage: telomeres, for example, shorten rapidly during the period of juvenile growth . On the other hand, many clinical biomarkers of ageing do not begin to accumulate until adulthood . This means that the applicability of biological-ageing measures to juveniles depends on the specific biomarkers being used, and different markers are likely to be most appropriate for measuring juvenile vs adult wellbeing .
Animals very different from humans
The evidence supporting the use of biological ageing markers as measures of cumulative welfare falls into three broad categories: a theoretical, evolutionary argument linking the affective status of an exposure to its effect on ageing via its effect on somatic damage; extensive empirical research in humans associating biological ageing markers with directly reported affective mood (e.g. stress, depression or anxiety) and experiences known to impact mood (e.g. trauma, pain, exercise, and sleep); and more limited empirical research in animals linking these markers to experiences that are both plausibly affectively relevant and associated with other widely-used welfare measures.
The second of these, empirical data on humans, is particularly important, as only humans are capable of directly reporting their affective state to human researchers and so directly confirming a link between biological ageing and welfare. As one moves away from humans in terms of the species under investigation, the less weight can be put on this source of evidence in support of this link, and the more one has to rely on the first and third sources of evidence outlined above. The more distant and dissimilar a species is from those species in which ageing-base techniques have been empirically studied, the more our confidence in those techniques should decrease towards the level of confidence we have in the theoretical argument alone. This poses an issue, since the vast majority of animals on the planet fall into this category.
There are two particular groups of animals for which I think the existing empirical data provides relatively little support for ageing-based welfare measures: invertebrates, and those animals (both vertebrate and invertebrate) whose pattern of lifetime ageing differs substantially from that of humans. The reasons for scepticism in the first case are clear: invertebrates are very different from vertebrates in many aspects of their biology, differ substantially from mammals in terms of their biomarkers of ageing (see above), are very diverse amongst themselves, and are almost totally unstudied as objects of welfare concern. My concerns about the second group, however, are likely to be less clear to someone outside the ageing field, and I will try to briefly explain my reasons below.
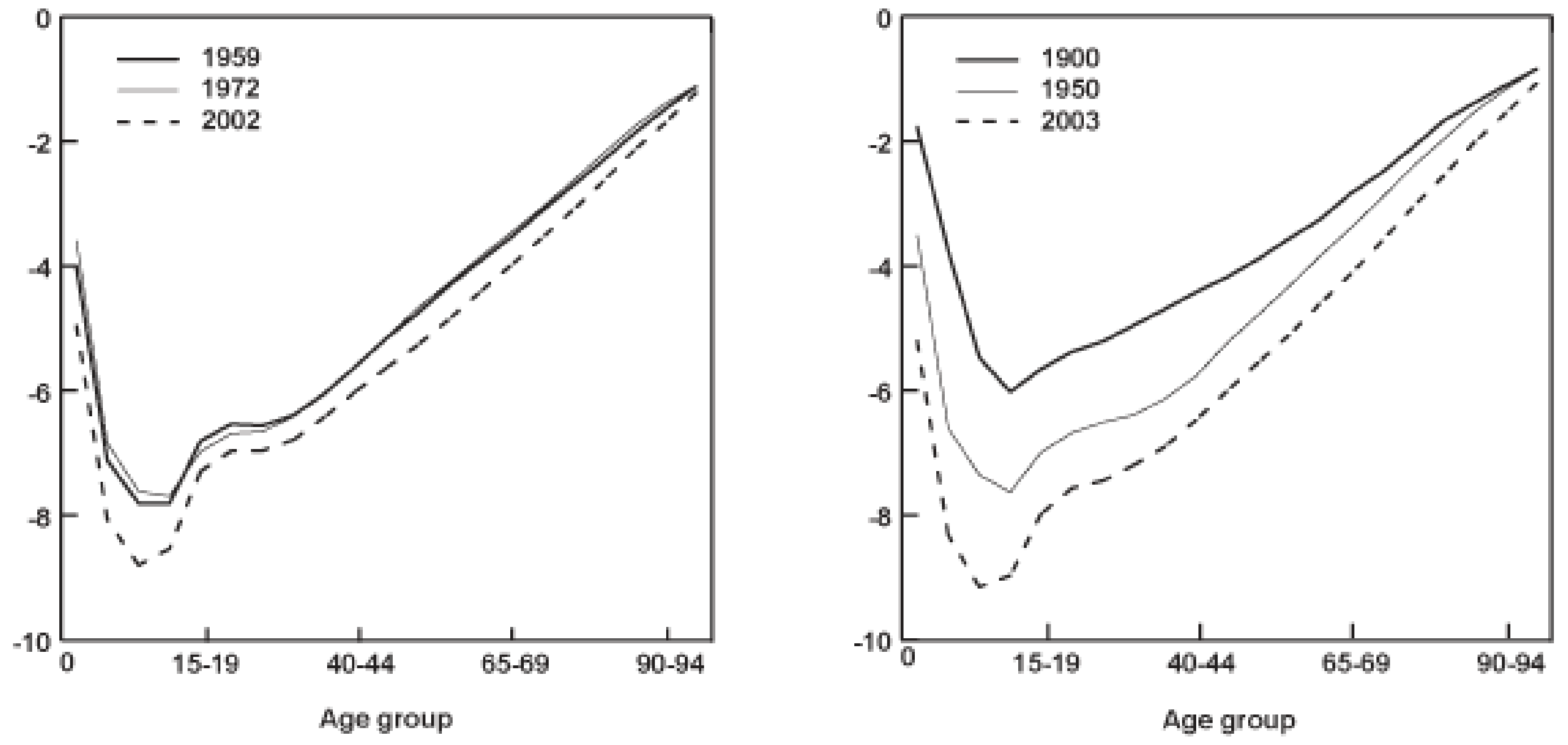
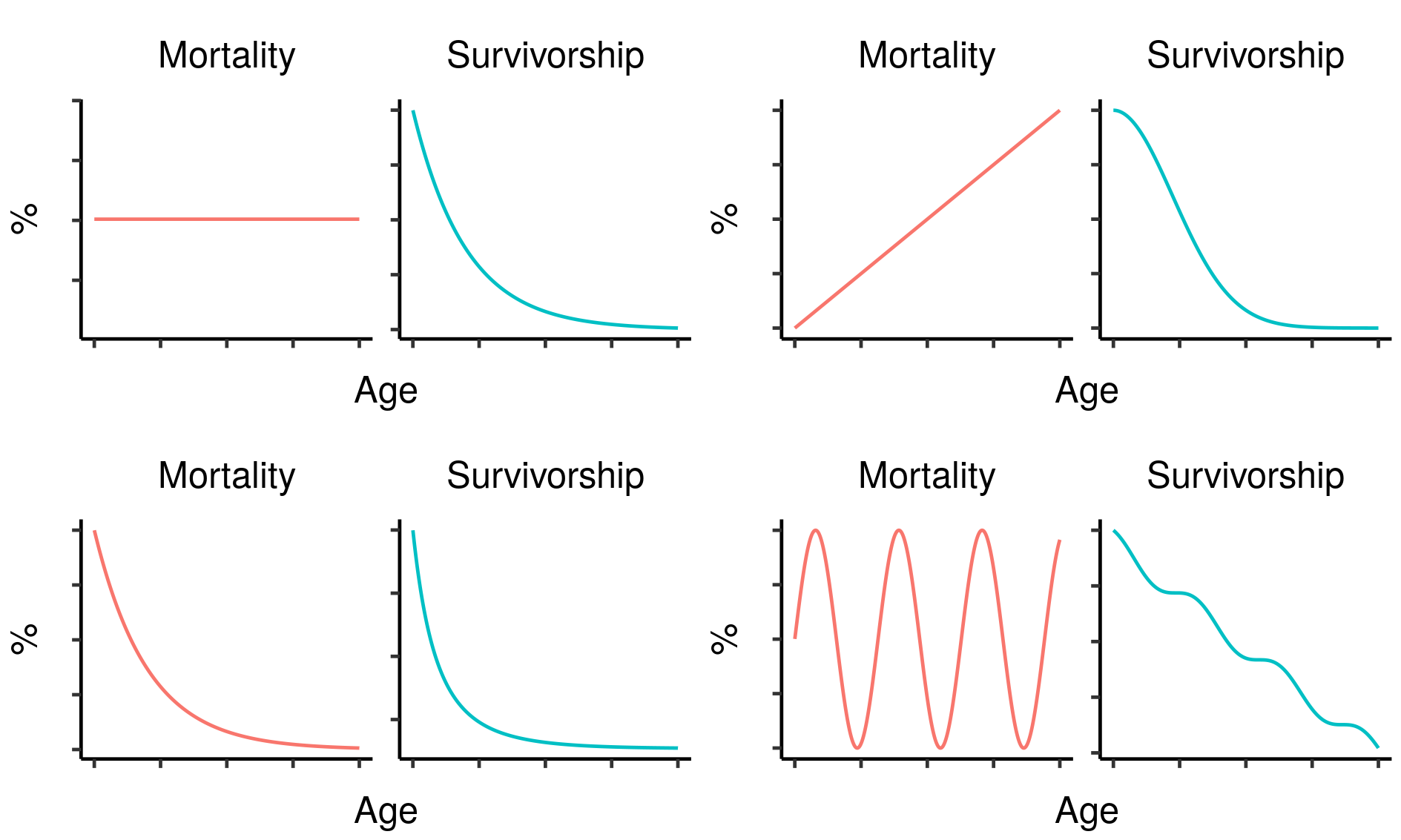
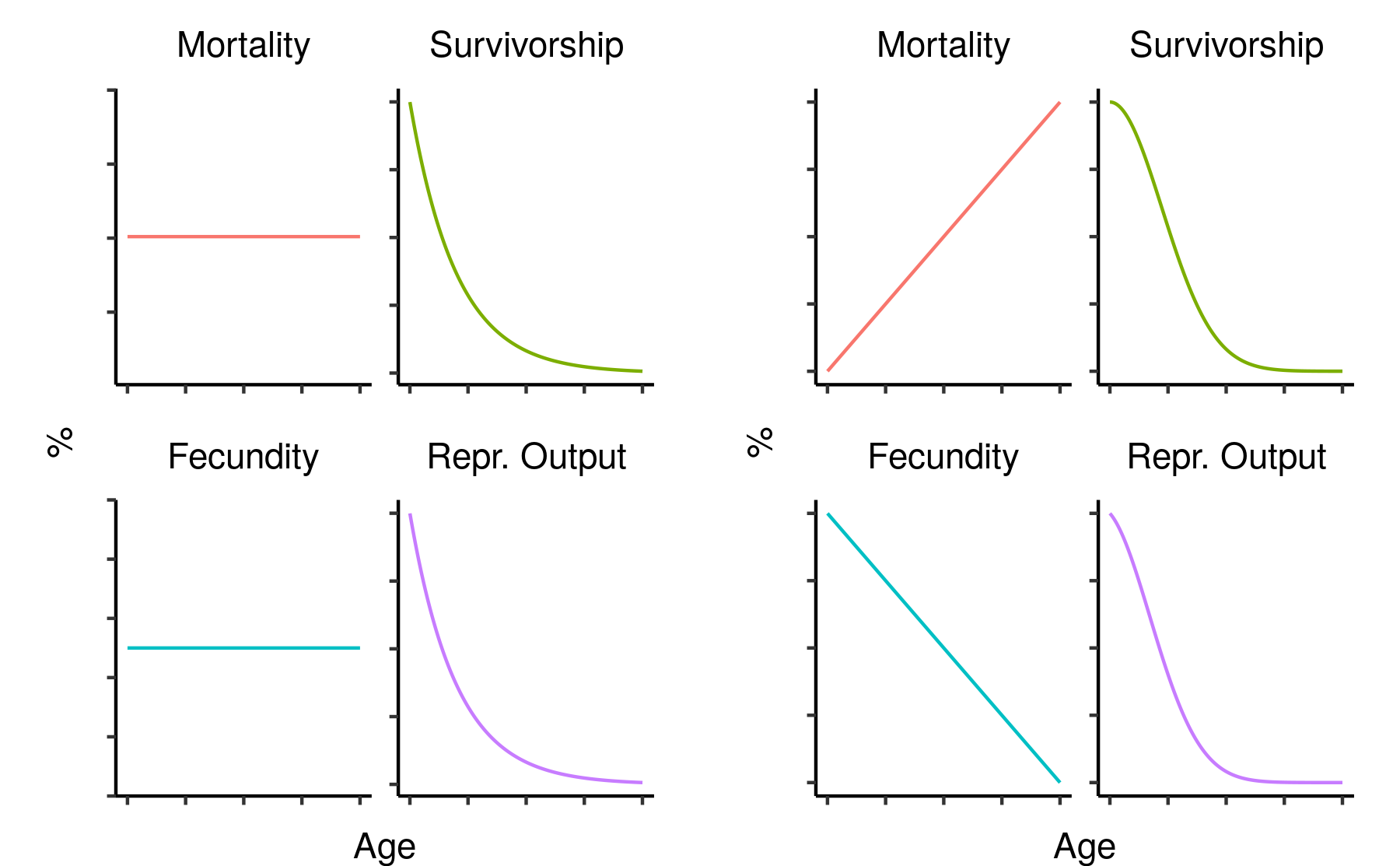
Typically, under simple assumptions that are frequently roughly met in real animals, we expect to see mortality progressively increase and fecundity progressively decrease with time after reproductive maturation . Many animals, including humans and nearly all common domesticated species, follow this pattern, but this does not apply universally to all animals. Some species, most famously the green hydra and more recently the naked mole rat , do not appear to age at all, while some (including various corals, reptiles and amphibians ) seem to “age in reverse”, exhibiting declining mortality with age until death.
These differences in life history could pose major problems for the use of biological ageing markers as a measure of cumulative welfare in these species. To begin with, it isn’t clear how to define the concept of biological age, let alone measure it, in an animal that does not age in any conventional way. Some subset of conventional biomarkers of ageing may still accumulate with time in these species, but that subset is likely to differ from taxon to taxon depending on what biological methods they have used to overcome the ageing process. Worse, even the theoretical argument in support of ageing-based welfare measures in these species may be greatly weakened: for an animal to exhibit no ageing or even reverse ageing over a prolonged time period, they must be either extremely resistant to somatic damage or have extremely good mechanisms in place to repair that damage, meaning the relationship between damaging (and therefore aversive) experiences and ageing may be largely or entirely severed.
These issues will pose little difficulty to researchers and activists concerned with the welfare of agricultural, experimental or other captive animals, most or all of which, to my knowledge, follow the conventional pattern of ageing exhibited by humans, mice and Drosophila. However, they could turn out to be significant for researchers interested in quantifying and improving the welfare of wild animals, who will inevitably have to tackle the welfare of large numbers of animals very different from humans. The extent to which these “unconventional” life histories are widespread in the natural world is unclear to me at present, and could be an important factor affecting the applicability of these methods in certain contexts.
Conclusion
Finding better methods with which to quantitatively measure the cumulative welfare experience of nonhuman animals would represent a major advance in the study of animal welfare. As a potential route to a better and more objective measure of cumulative welfare, biomarkers of ageing are potentially very promising.
Many of the caveats I outlined above are educated guesses and may well turn out to be circumventable with sufficient thought and care. If they are not, how serious a problem would this pose for the practical usefulness of this method? In the case of domesticated animals, an inability to compare groups differing systematically in their genetics or adequately incorporate the badness of death are all significant limitations, but would still leave us with a tool which could be gainfully applied in many important contexts. For wild animals, the issues are more serious: there are vastly more species for which we would have to develop methods of measuring biological age, it is much harder to perform well-controlled longitudinal experiments, and many more of the animals of concern fall into categories for which I am more sceptical about the theoretical applicability of the method. A further concern is that measuring chronological age accurately is often difficult for wild animals, potentially undermining one of the foundations of ageing-based welfare measures. Nevertheless, of all the methods we might think of for measuring the cumulative welfare of wild animals, biomarkers of ageing seem to be among the least hopeless, and among those most worth developing further in the hopes of overcoming some of these pervasive issues.
Overall, I am currently very optimistic about the value of applying these methods in domesticated contexts, and cautiously optimistic about applying them to wild ecosystems. I would recommend that researchers interested in the welfare of either domesticated or wild animals take note of these techniques and consider their applicability in their own domains, and that funders in this space seriously consider funding their further development and application to new contexts. On a meta level, I think the surprisingly strong applicability of techniques from the biology of ageing to animal welfare science should encourage us all to look for innovative, unexpected and interdisciplinary ways to help nonhuman animals: as is often the case in science, the crucial insights and discoveries may not be at all where we expect them to be.
Acknowledgements
This report was funded by the Wild Animal Initiative, and many WAI researchers (including Luke Hecht, Michelle Graham, Hollis Howe and Jane Capozzelli) contributed their time to reviewing and suggesting improvements to the draft. Prof. Melissa Bateson, the author of the key sources for the report, also generously read the draft and provided crucial feedback.
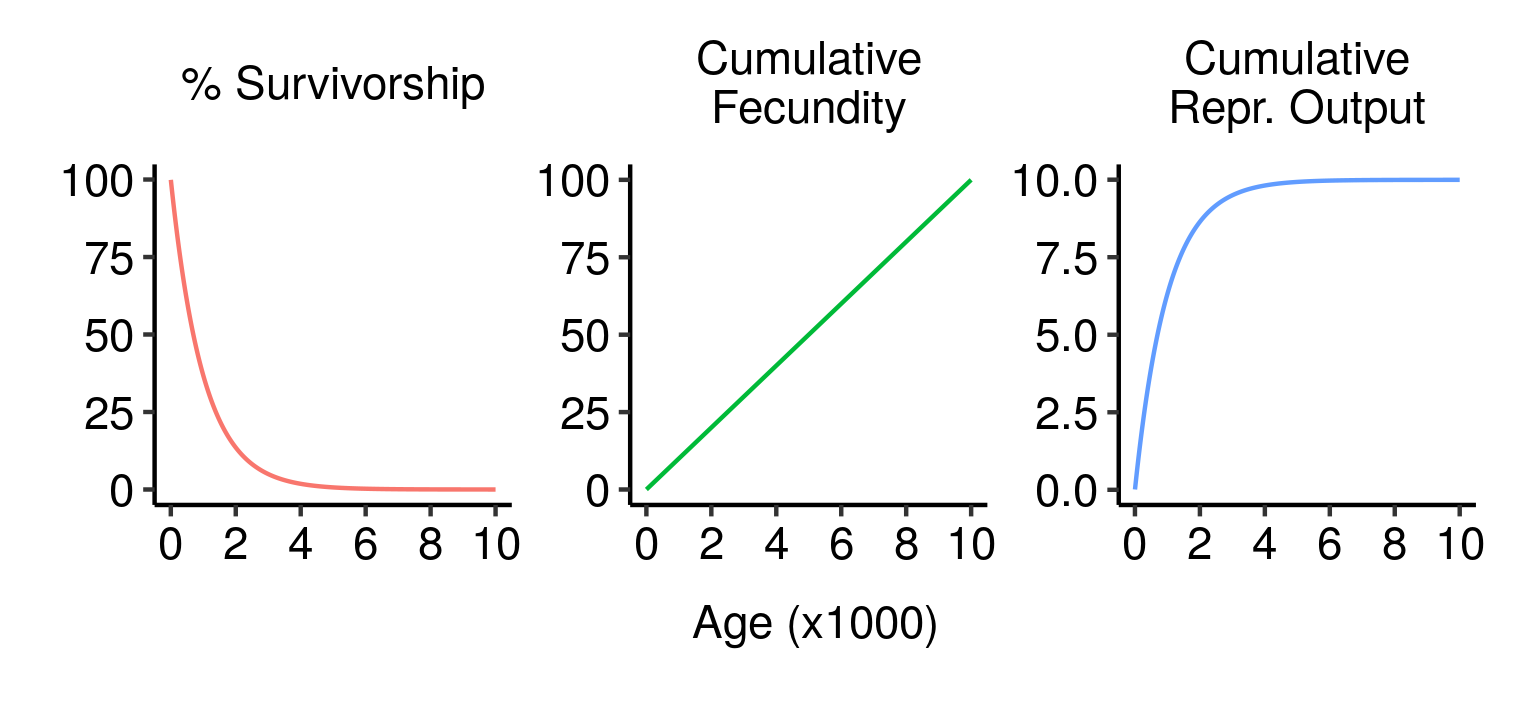 Survivorship, cumulative fecundity and cumulative reproductive output curves for a population of elves with 1% fecundity and 0.1% mortality per year.
Survivorship, cumulative fecundity and cumulative reproductive output curves for a population of elves with 1% fecundity and 0.1% mortality per year.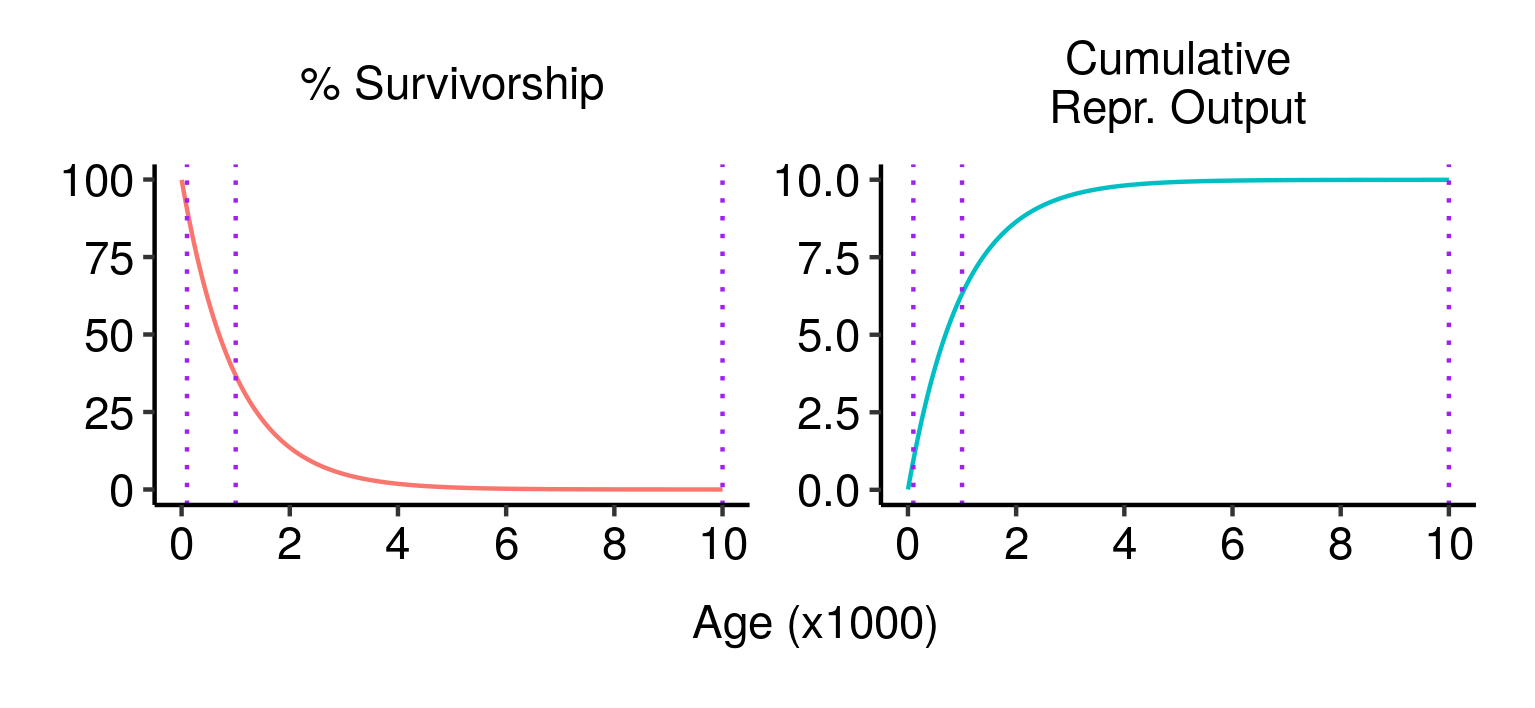 Three potential fatal mutations in the elven populations, and their effects on lifetime reproductive output.
Three potential fatal mutations in the elven populations, and their effects on lifetime reproductive output.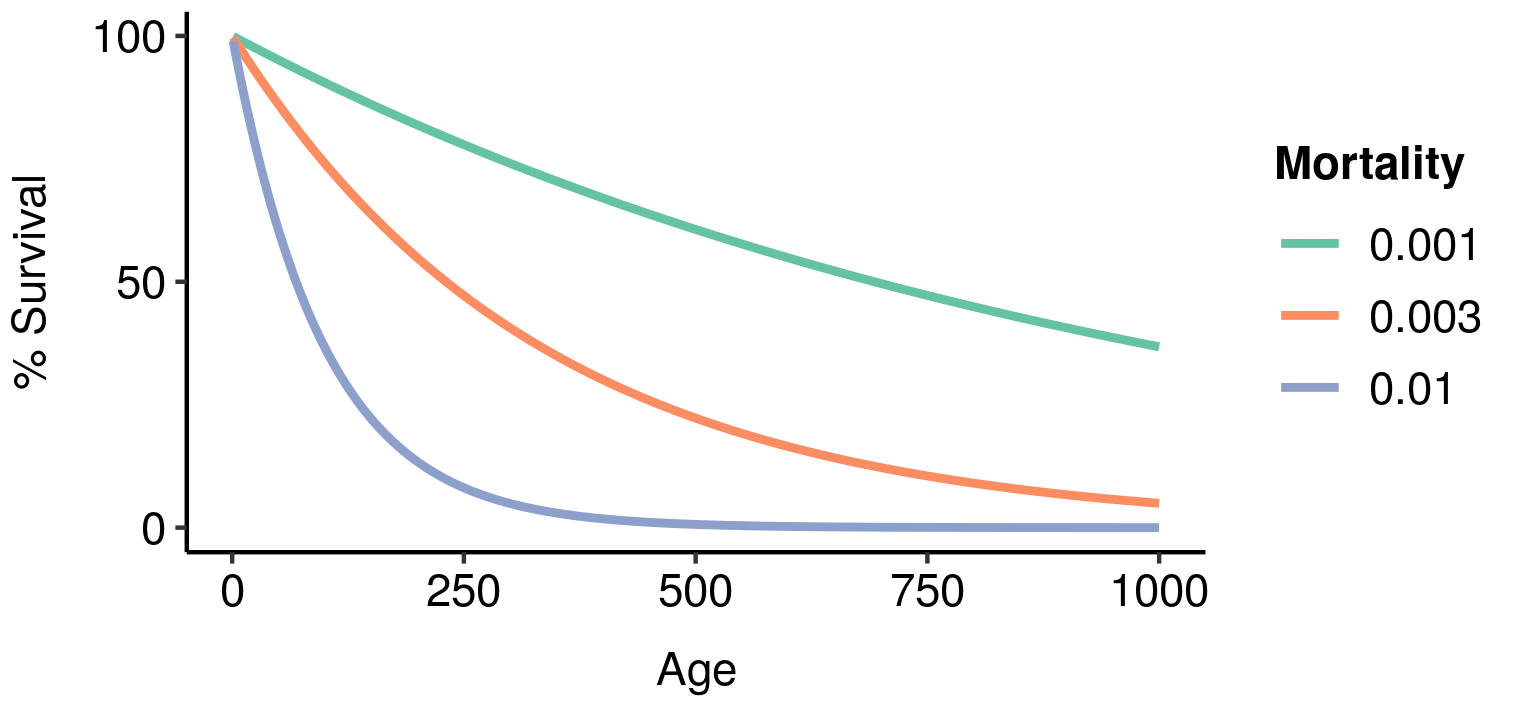 Age-specific survivorship as a function of different levels of constant extrinsic mortality.
Age-specific survivorship as a function of different levels of constant extrinsic mortality.
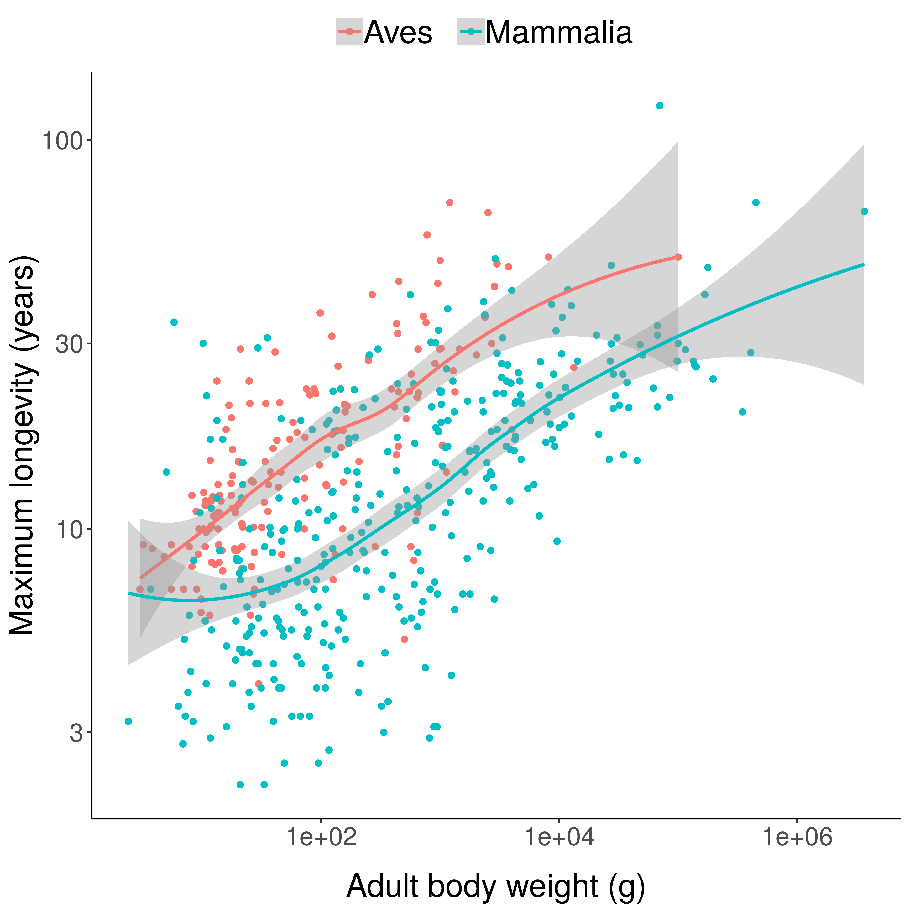 Scatterplots of bird and mammal maximum lifespans vs adult body weight from the AnAge database, with central tendencies fit in R using local polynomial regression (
Scatterplots of bird and mammal maximum lifespans vs adult body weight from the AnAge database, with central tendencies fit in R using local polynomial regression (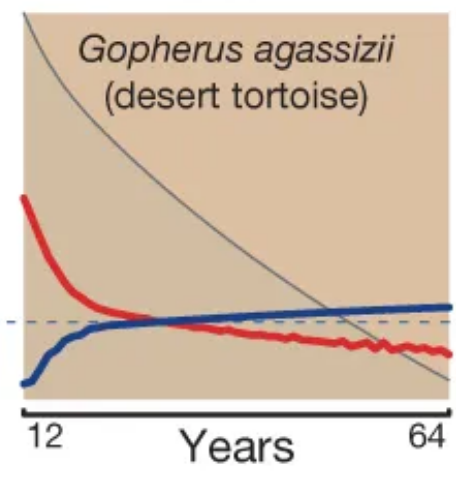 Mortality (red) and fertility (blue) curves from the desert tortoise, showing declining mortality with time. Adapted from Fig. 1 of
Mortality (red) and fertility (blue) curves from the desert tortoise, showing declining mortality with time. Adapted from Fig. 1 of